 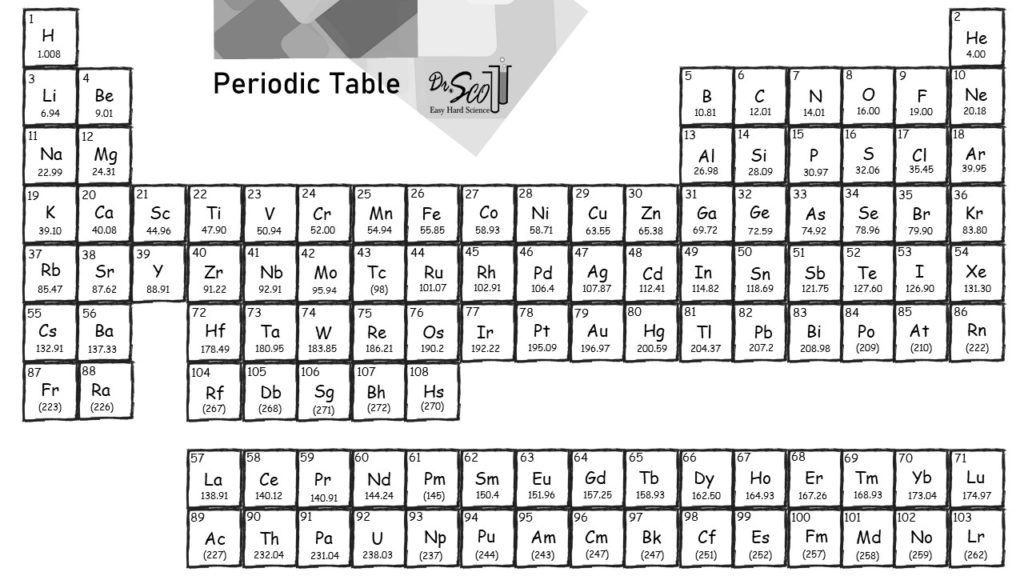
These individual element summary pages contain a lot of additional. When exploring the table or list views on this page, please note the links to dedicated pages for each element. CO 2 has one carbon atom and two oxygen atoms. PubChem is providing this periodic table page in order to help navigate abundant chemical element data available in PubChem.Add together the atomic masses of all of the atoms of hydrogen and water in a molecule of water : 2 x 1.008 g (hydrogen) + 1 x 16.00 g (oxygen) 18.02 g. The molar mass of water is the mass of one mole of H 2 O. Oxygen (O) has an atomic mass of about 16.00 amu. Apply the same principles to calculate the molar mass of a molecule.Carbon (C) has an atomic mass of about 12.01 amu.Let's calculate the molar mass of carbon dioxide (CO 2): Add them together: add the results from step 3 to get the total molar mass of the compound.Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound.The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). Find atomic masses: look up the atomic masses of each element present in the compound.For example, water is H 2O, meaning it contains two hydrogen atoms and one oxygen atom. Identify the compound: write down the chemical formula of the compound.One mole contains exactly 6.022 ×10 23 particles (Avogadro's number) Mole is a standard scientific unit for measuring large quantities of very small entities such as atoms and molecules.Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol.(1 u is equal to 1/12 the mass of one atom of carbon-12) Molecular mass ( molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u).To calculate molecular weight of a chemical compound enter it's formula, specify its isotope mass number after each element in square brackets.Įxamples of molecular weight computations: Molar mass calculator also displays common compound name, Hill formula, elemental composition, mass percent composition, atomic percent compositions and allows to convert from weight to number of moles and vice versa.Ĭomputing molecular weight (molecular mass) Functional groups: D, Ph, Me, Et, Bu, AcAc, For, Tos, Bz, TMS, tBu, Bzl, Bn, DmgĮxamples of molar mass computations: NaCl, Ca(OH)2, K4, CuSO4*5H2O, nitric acid, potassium permanganate, ethanol, fructose, caffeine, water.Capitalize the first letter in chemical symbol and use lower case for the remaining letters: Ca, Fe, Mg, Mn, S, O, H, C, N, Na, K, Cl, Al. Nitrogen is a colorless, odorless gas that exists as diatomic molecules (N2) in its most common form.Ĭomputing molar mass (molar weight)To calculate molar mass of a chemical compound enter its formula and click 'Compute'. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
